Project 1's objective is to characterize several novel transcription factors, not previously implicated in addiction, in mediating the lasting actions of stimulant and opioid drugs of abuse in nucleus accumbens (NAc) and dorsal striatum (DS). Virtually all prior studies of transcription factors in addiction have taken a candidate approach. This is in marked contrast to an unbiased approach of using “big data” to deduce, in an open-ended manner, those factors that are most important in mediating specific aspects of the complex addiction phenotype. We utilize this more powerful approach by taking advantage of large-scale RNA-seq and related datasets from rodent addiction models and humans with substance use disorders to identify those factors that appear to play particularly critical roles in drug action. We focus on cell type-specific actions of three of the most highly ranked transcription factors from our PPG’s datasets: E2F3, ZFP189, and RXR. For example, ~25% of all genes that display primed or desensitized changes in expression in NAc as a result of past cocaine self-administration are predicted to be direct targets of E2F3. Based solely on these bioinformatics predictions, we have generated robust preliminary data to validate the importance of each of these transcription factors in cocaine and opioid action, and now propose to better understand their actions. We will complete characterization of their role in NAc in controlling behavioral responses to cocaine in self-administration assays as well as map their target genes on a genome-wide basis. Both of these efforts will be performed in a cell type-specific manner, as we have evidence for some of the factors playing different roles in different neuronal types in this brain region. This work in animal models will be complemented by studies of humans with cocaine use disorders, where we already have preliminary evidence for their abnormal regulation. We will extend these studies to opioid models where we have increasing evidence for each of these three transcription factors also showing prominent dysregulation in NAc. As well, we will explore their involvement in DS. Together, this work will reveal new transcriptional mechanisms underlying cocaine and opioid addiction.
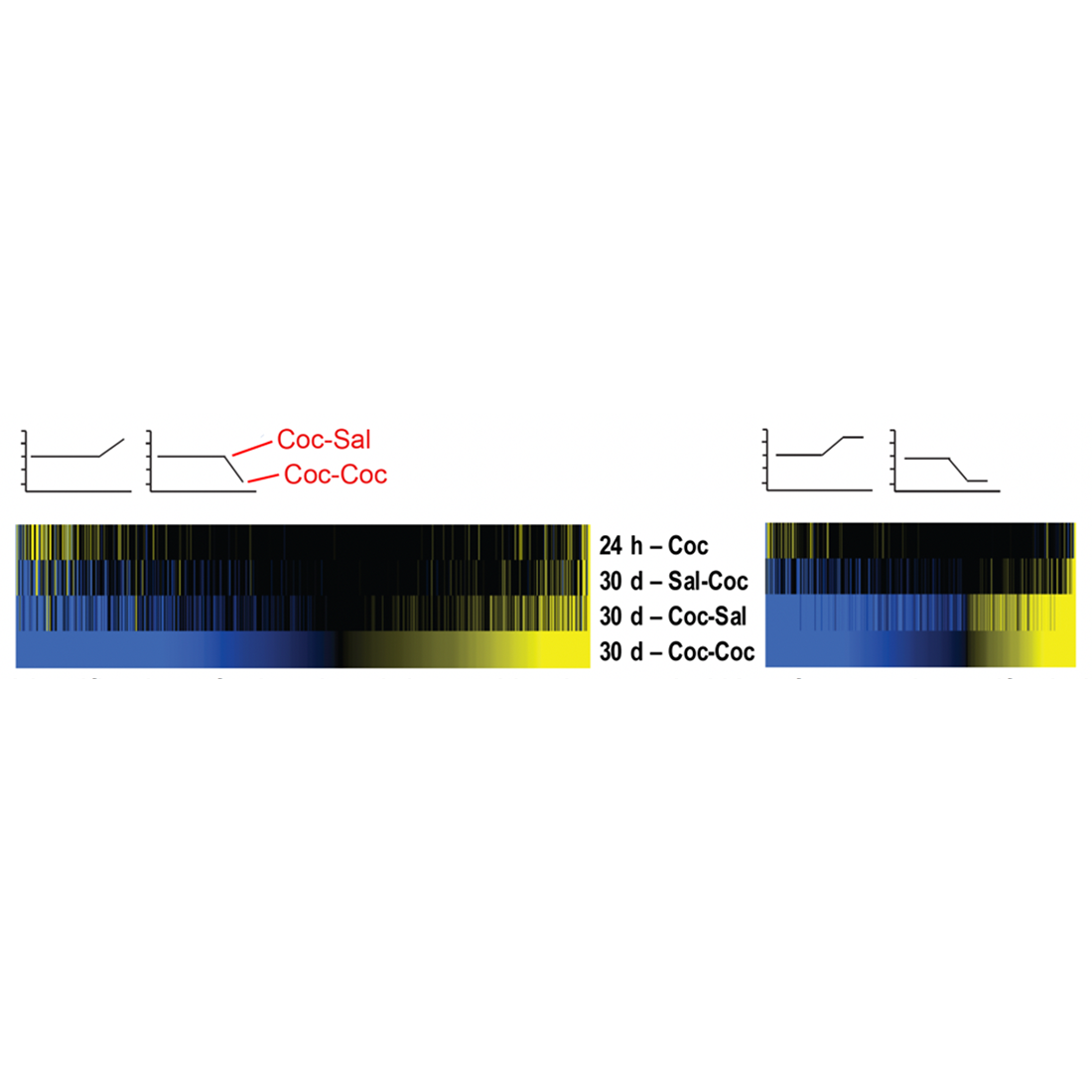
Identification of primed and desensitized genes in NAc after cocaine self-administration. See Walker et al., Biol Psychiatry, 2018.