Project 3's objective is to address the role of microglia in modulating transcriptional adaptations in nucleus accumbens (NAc) and dorsal striatum (DS) medium spiny neurons (MSNs) triggered by cocaine or opioid self-administration. Microglia contribute to normal brain development and function by supporting neuronal survival and removing non-functional neurons and synapses. We found that ablation of microglia increases behavioral responses to cocaine, to morphine, and to a D1 dopamine receptor agonist; such ablation also increases dendritic spine density on NAc and DS MSNs. These findings suggest the hypothesis that microglia may function homeostatically to oppose conditions of excessive dopaminergic transmission, such as seen with cocaine or opioid exposure. We have found as well that 10-15% of microglia in striatum, but not other brain regions, express the D1 receptor, raising the novel possibility of direct effects of drug (via increased dopaminergic transmission) on microglia. We now propose to fully characterize the influence of microglia in controlling behavioral responses to cocaine and opioids in self-administration models. This will include delineating a role for all microglia in striatum as well as a possible selective role played by D1+ microglia and by D1 receptor signaling within those microglia. We will next characterize the influence of microglia—again D1+ and D1- subpopulations—on the ability of cocaine or opioid self-administration to influence gene expression profiles in the D1-type and D2-type MSNs of NAc and DS. We will also characterize the changes in gene expression induced by cocaine or opioid self-administration in D1+ and D1- subpopulations of microglia themselves within striatum. These experiments are made possible by several novel lines of genetic mutant mice that enable the selective manipulation of microglial subpopulations within striatum combined with RNA-seq of isolated neuronal and microglia cell types—and even at the single cell level. These investigations of cocaine and opioids in mice set the stage for follow up studies of humans with substance use disorders. Overall, the proposal has the potential to provide paradigm-shifting information about the role of microglia in the pathophysiology of drug addiction.
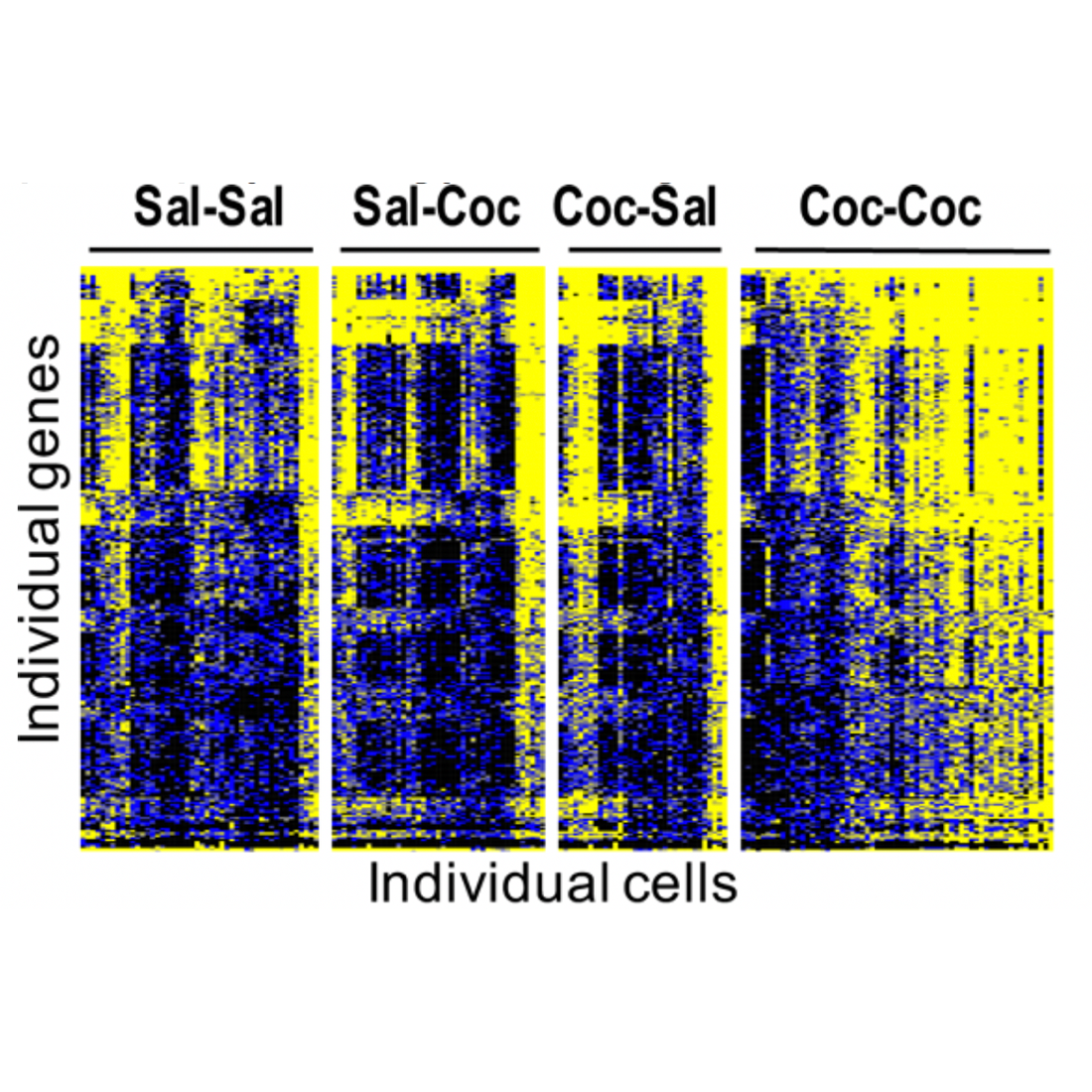
Single cell RNA-seq of microglia reveals gene regulation by cocaine self-administration.